
Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (C 12 H 22 O 11 (sucrose)). Then, lookup atomic weights for each element in periodic table: C: 12.0107, H: 1.00794, O: 15.9994. The molar mass of a compound is the mass in grams of 1 mole of that substance, a property expressed in units of grams per mole (g/mol)īecause the definitions of both the mole and the formula mass are based on the same reference substance, 12C, the molar mass of any compound is numerically equivalent to its formula weight in amu. First, compute the number of each atom in C 12 H 22 O 11 (sucrose): C: 12, H: 22, O: 11. The original periodic table of the elements published by Dimitri Mendeleev in 1869 arranged the elements that were known at the time in order of increasing atomic weight, since this was prior to. The masses of 1 mole of different compunds, however, are different, since the masses of the individual molecules are drastically different. One molecule of water (H 2 O) would weigh 18.02 amu (2×1.00797 amu for H + 15.9994 amu for O), and a mole of water molecules would weigh 18.02 grams. The calculated value makes sense because it is almost four times times the mass for 1 mole of aluminum. Find your chosen element on the periodic. Prepare a concept map and use the proper conversion factor. A property closely related to an atom’s mass number is its atomic mass. If you want to calculate how many neutrons an atom has, you can simply subtract the number of protons, or atomic number, from the mass number. Locate the element on the periodic table: The periodic table is arranged systematically and provides essential information about elements such as atomic numbers (number of protons) and atomic weights. Together, the number of protons and the number of neutrons determine an element’s mass number: mass number protons + neutrons. 1: Moles to Mass Conversion with Elements. According to the periodic table, 1 mol of U has a mass of 238.0 g, so the mass of 2 mol is twice that, or 476.0 g. Understanding these furniture is crucial for comprehending the realm of scientific research. See also our theoretical yield calculator for chemical reactions (probably your next stop to finish the problem set).
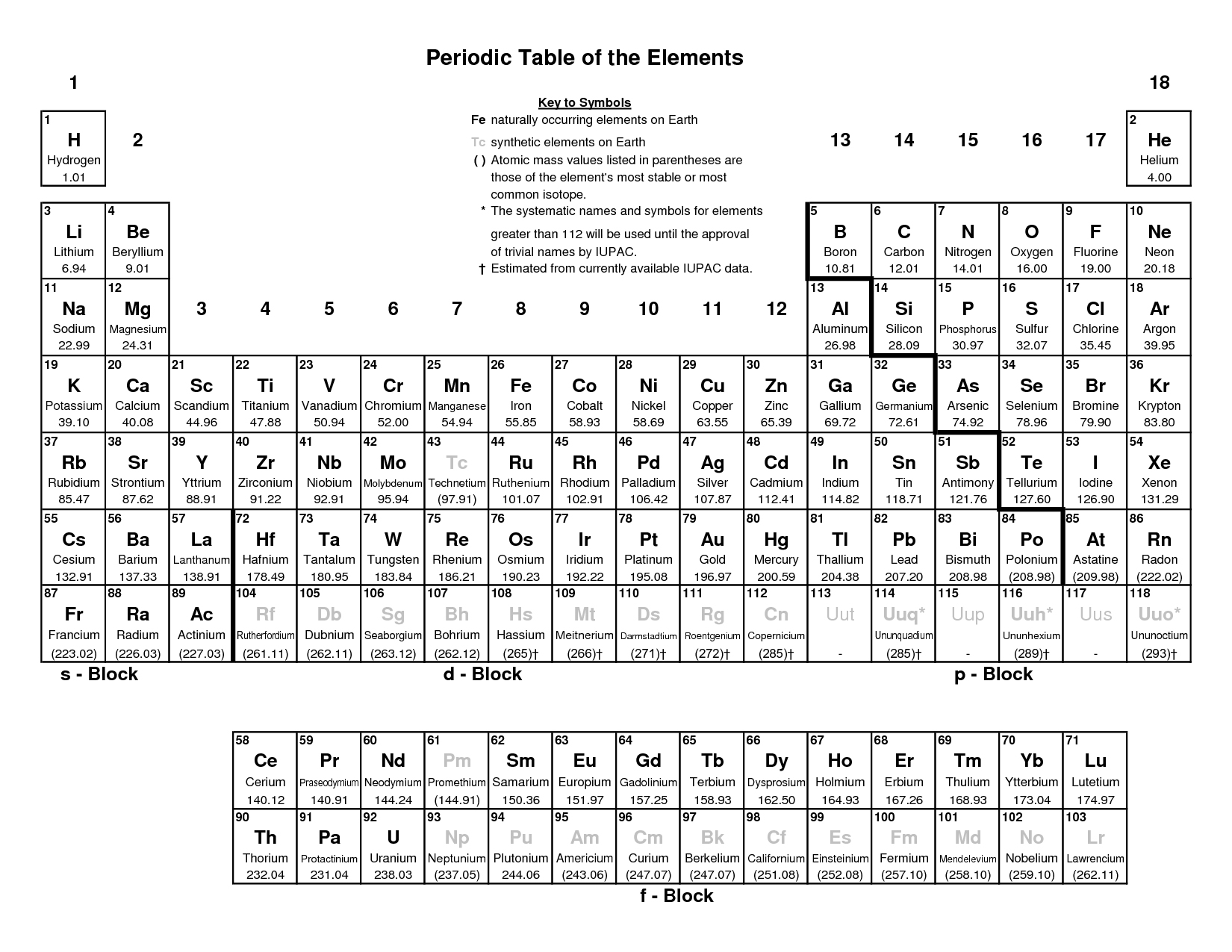
Here are a few samples of regular furniture and their features. The molar mass of NO2 (Nitrogen dioxide) is: 46.005 grams/mol. Similar to atoms, 1 mole of any compound contains the same number of molecules as 1 mole of any other compund. For example, if we want to find the molar mass of carbon, we can use the elemental symbol ‘C’. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. In terms of mass, a mole (mol) is the relative atomic mass close relative atomic mass The mean relative mass of the atoms of the different isotopes in an element. This technique is very simple, and doesn’t demand testing.
