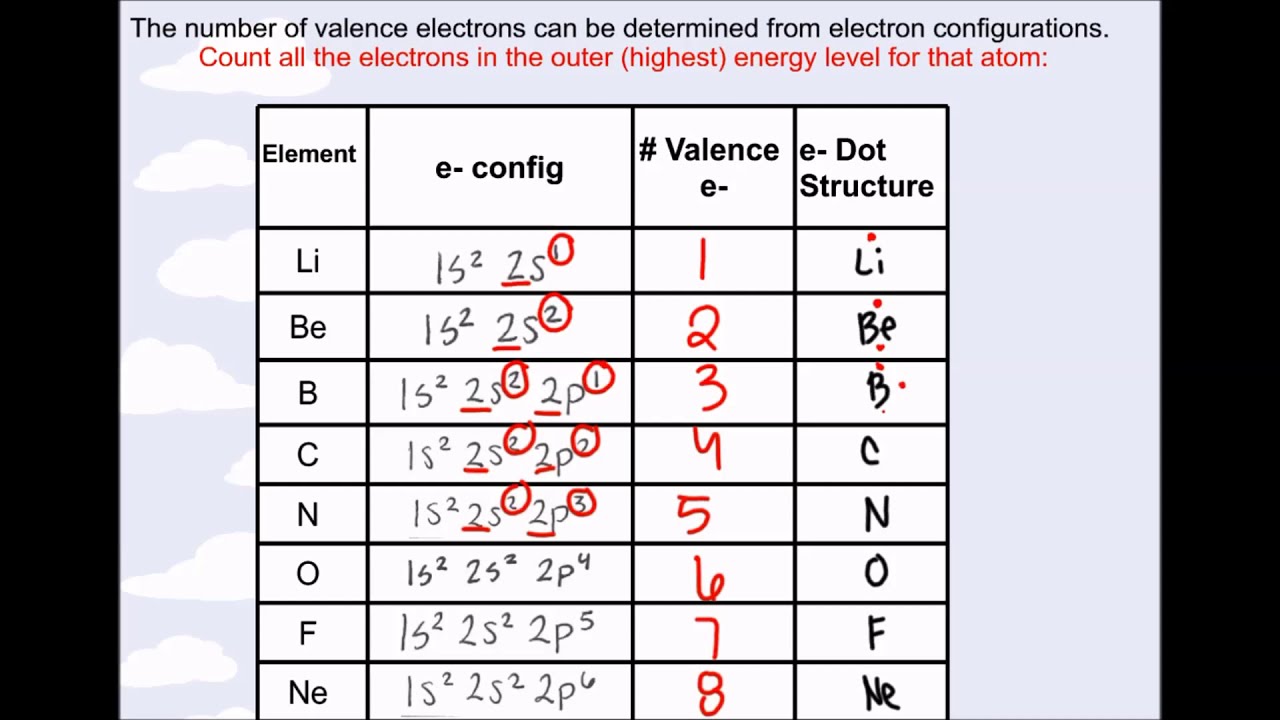
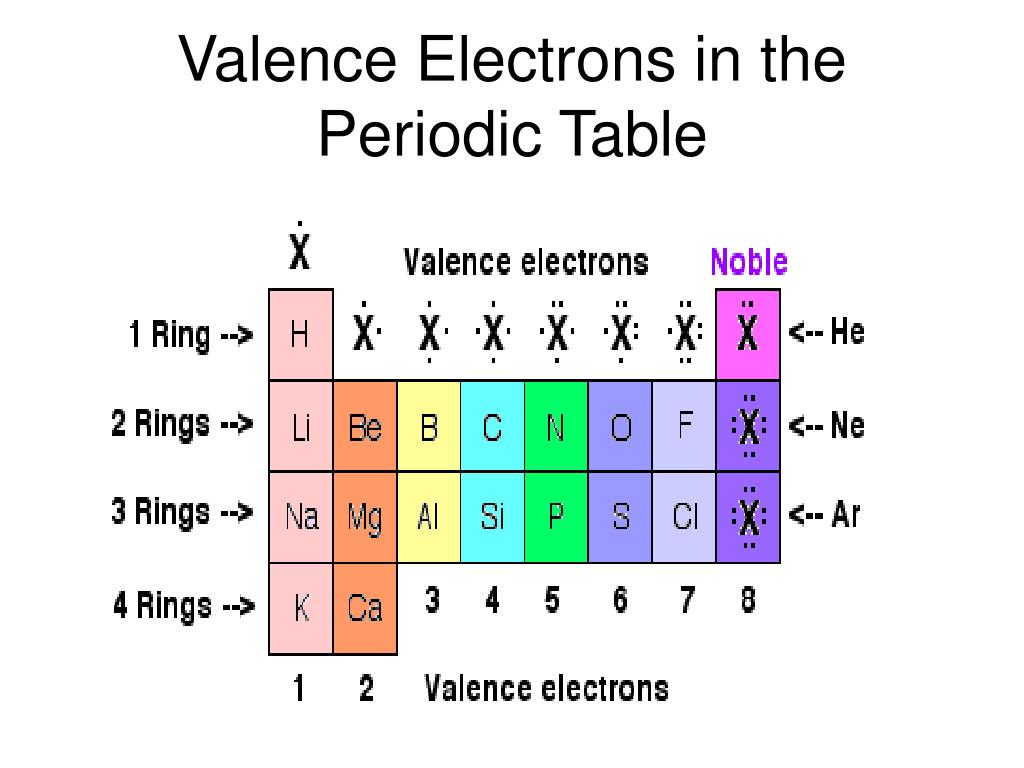
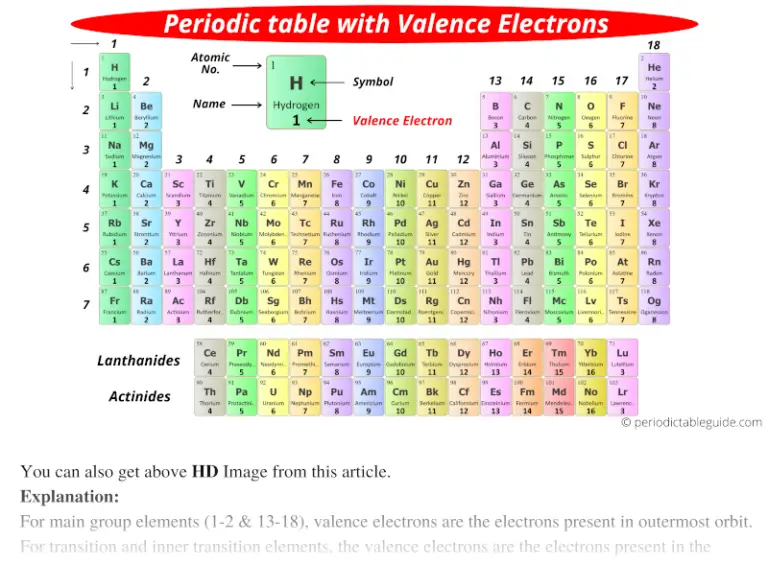
Basically, atoms are most stable when they can achieve an outer electron structure similar to the closest Noble gas.This can be done by either sharing electrons between atoms, taking electrons from other atoms, or giving electrons away to other atoms. If atoms don't have 8 valence electrons (or two if they are close to He), then they will react with other atoms in order to have 8 valence electrons.One exception is He which has a full principle energy level with 2 electrons. All of these elements have 8 valence electrons in their highest principle energy level. One simple piece of evidence for this is the Noble Gases which form the last column on the right of the periodic table.
Hemoglobin is a relatively large molecule, with a mass of about 65,000 u. Hemoglobin combines with oxygen and carbon dioxide, transporting these gases from one location to another in the body. The critical part of the red blood cell is a protein called hemoglobin. Without red blood cells, animal respiration as we know it would not exist. Red blood cells are cells that transport oxygen from the lungs to cells of the body and then transport carbon dioxide from the cells to the lungs. The chemistry of iron makes it a key component in the proper functioning of red blood cells.
Transition metals have interesting chemical properties, partially because some of their electrons are in d subshells. Because iron has relatively massive atoms, it would appear even lower on a list organized in terms of percent by atoms rather than percent by mass. The first element appearing on the list that is not a main group element is iron, at 0.006 percentage by mass. Most of the elemental composition of the human body consists of main group elements. To Your Health: Transition Metals in the Body Each group is located in a different part of the periodic table. Elements are either metals, nonmetals, or semimetals.
